Nanospecialists have completed a review of the 2D nanomaterial borophene, finding it to be a material with exceptional properties and greater potential than graphene.
The report has now been published in the journal Frontiers of Physics, where they state, “Research on borophene has grown fast in the fields of condensed matter physics, chemistry, material science, and nanotechnology. Due to the unique physical and chemical properties, borophene has various potential applications. In this review, we summarize the progress on borophene with a particular emphasis on the recent advances.”
The study was conducted by an international team from Xiamen University in China and the National University of Singapore, and explored not only borophene’s noteworthy abilities, but also its potential applications in industry. Here is a summary of its contents.
The Short History of Borophene.
When graphene was discovered in the mid-1990’s it was instantly hailed as a new ‘wonder material’. Measuring only one atom thick, it was the first truly 2D material. Graphene is made from an arrangement of graphite (used to make objects such as pencil ‘lead’) into a hexagonal lattice, but it is its technical properties that amazed the scientific and industrial world.
As the graphene news portal Graphene-Info explains, “Graphene is the thinnest material known to man at one atom thick, and also incredibly strong - about 200 times stronger than steel. On top of that, graphene is an excellent conductor of heat and electricity and has interesting light absorption abilities. It is truly a material that could change the world, with unlimited potential for integration in almost any industry.”
As a result, investment from business and government quickly flooded in to take advantage, including a €1 billion injection from the EU to kick-start a graphene industry.
While there has been a return on this investment, it has trickled in rather than the expected tsunami, and today supports a healthy, productive graphene community of firms.
But perhaps graphene’s greatest achievement has been to fuel interest into other 2D materials, with research ongoing in germanene, graphyne, silicene, plumbene, and stanine, to name a few.
Most curious of all is borophene: a single layer of boron atoms in various crystalline structures.
First predicted by computer simulations in the 1990’s, borophene didn’t actually come into existence until as recently as 2015.
Borophene Production
Like many nanomaterials, borophene is synthesized using a process of chemical vapor deposition. This involves heating boron atoms into a hot gas which is then left to condense onto a cool surface of pure silver.
It is the relationship between the boron and silver atoms that makes borophene exceptional. As the scientific journal Technology Review, describes, “The regular arrangement of silver atoms forces boron atoms into a similar pattern, each binding to as many as six other atoms to create a flat hexagonal structure. However, a significant proportion of boron atoms bind only with four or five other atoms, and this creates vacancies in the structure. The pattern of vacancies is what gives borophene crystals their unique properties.”
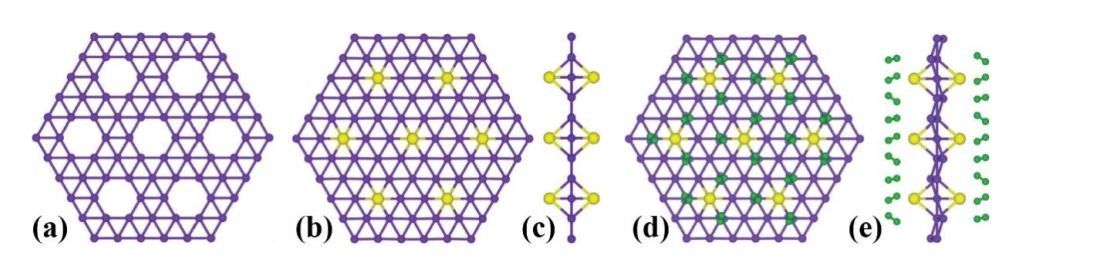
Borophene; a Nanomaterial for Many Industries
The real excitement over borophene is the potential it holds in so many valuable sectors of industry. Stronger and more flexible than graphene, borophene has possible applications in catalytic reactions in the chemical industry, could be used in producing high-end sensors and testing equipment for detecting various molecules and atoms, or as an anode material for next-generation lithium-ion batteries.
As Zhi-Qiang Wang, a professor at Xiamen University in China who led the study, highlights, “Borophene is a promising anode material for Li, Na, and Mg ion batteries due to high theoretical specific capacities, excellent electronic conductivity and outstanding ion transport properties.”
Borophene also has a special relationship with hydrogen, as Technology Review, states, “Hydrogen atoms also stick easily to borophene’s single-layer structure, and this adsorption property, combined with the huge surface area of atomic layers, makes borophene a promising material for hydrogen storage. Theoretical studies suggest borophene could store over 15% of its weight in hydrogen, significantly outperforming other materials.”
Additionally, chemists are keen to explore borophene’s ability to catalyze the breakdown of molecular hydrogen into hydrogen ions, and water into hydrogen and oxygen ions. Properties that could lead to discoveries in water-based energies.
As the review explains, “The outstanding catalytic performances of borophene have been found in hydrogen evolution reaction, oxygen reduction reaction, oxygen evolution reaction, and CO2 electroreduction reaction.”
Borophene is also light and reasonably reactive, making it a suitable candidate for storing metal ions in batteries. It is also a good conductor of heat and electricity, and can also act as a superconductor.
All of these properties are dependent on the orientation and the arrangement of vacancies on the 2D structure. This makes borophene, at least in theory, ‘tuneable’, with nanospecialists able to vary its production to enhance or shutdown various qualities.
Borophene’s Future
As borophene is such a new material, there is still plenty of research to be done before nanotechnology can fully realize its potential. Industrial scale production has yet to be economically achieved, and the material’s reactivity means it is vulnerable to oxidation, so care must be taken in handling to ensure it is properly protected.
Both of these factors have been overcome with other nanomaterial products, leaving borophene with a hopeful future.
Graphene’s labelling as a ‘wonder material’, the large amount of investment it has received, and the perception that it has failed to live up to expectations, has left many considering if nanomaterials will truly impact the way that we live. Yet one only has to examine the long list of applications for nanowires, nanosheets, nanoparticles, and nanotubes, and how they are already being used to see that nanotechnology is changing our world.
While today, it would be correct to see graphene as only a small part of the nano-industrial revolution, borophene’s even greater list of abilities and potential uses makes it a raw material worthy of attention.
Photo credit: Grapheneinfo, & Phys.org
